Unit 6: Chemical Bonding and Nomenclature
Objectives:
Lewis Structures
- covalent compounds
- acids and bases
- hydrates
covalent compounds
acids and bases
hydrates
Lewis Structures
- express the arrangement of valence electrons in an atom using a Lewis, valence electron, dot structure.
- name the following types of compounds:
- covalent compounds
- acids and bases
- hydrates
- write formulas of common polyatomic ions
- write formulas of the following types of compounds:
covalent compounds
acids and bases
hydrates
- describe metallic bonding
- explain metallic properties such as thermal and electrical conductivity, malleability, and ductility in terms of the metallic bonding theory
- Explain VSEPR theory
- Predict the shapes of molecules or polyatomic ions using VSEPR theory.
- Explain what determines polarity
Unit 6 Packet - Chemical Bonding and Nomenclature
Day 0 |
Day 6.1 2/22 |
Day 6.2 2/24 |
|
What is an ionic bond and how do I write a formula? Please review and take notes over the following ionic bonding tutorial. The course code is 95773077. Please take complete the practice problems afterwards and submit a picture in goformative.
Chemthink.com Ionic Bonding Tutorial goformative.com |
hat is an ionic bond and how do I write a formula? Today we will begin to explore ionic bonds and how they form. We will also begin looking into how to create formulas for ionic compounds.
Please review and take notes over the following ionic formulas tutorial from simbucket.com Once you have completed the tutorial and problem set please upload an image of your score to goformative.com. Tools to help memorize the ions. |
How do I name an ionic compound? Ionic compounds are formed due to the electrostatic attraction of a positive ion to a negative ion. The overall compound is electrically neutral and contains enough of each ion to balance the charges. Naming conventions were developed by IUPAC and the basic rule for naming ionic compounds is as follows:
Please review and take notes over the covalent bonding tutorial from simbucket.com Once you have completed the tutorial and problem set please upload an image of your score to goformative.com. |
Day 6.3 2/27 |
Day 6.4 3/1 |
Day 6.5 3/6 |
|
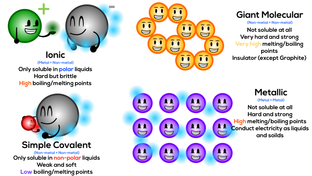
Metallic and Covalent Bonding, Covalent, Acid, and Hydrate Nomenclature: Today we will discuss the difference between metallic, covalent, and ionic bonds. How to name covalent molecules, acids and hydrates. Remember covalents are the only one with prefixes.
Metallic & Covalent Bonding |
Practice Everything Naming: Today we will be practicing naming everything. You are required to earn a maximum of 30 points from the activities provided. Your score will be based on the points earned.
Bonding and Nomenclature Activity Sheet Nomenclature Activity Review Nomenclature Activity Solutions HodgePodge Naming review HodgePodge Solutions Properties and Naming Foldable Properties and Naming Solutions |
Quiz: Quiz covering nomenclature and bonding
What Shape do the Molecules Make: Today we will be determining the structure and drawing the molecules using Lewis Structures (aka electron dot structures). Please review and take notes over the following molecular shapes tutorial from simbucket.com Once you have completed the tutorial and problem set please upload an image of your score to goformative.com. |
Day 6.6 3/8 |
Day 6.7 3/10 |
Day 6.8 3/20 |
|
How do molecules look in 3 dimension? Today we will discover how to 3D optimize Lewis Structures using VSEPR theory.
|
Today we will discuss and practice how to calcualte the percent composition by mass of a compound. Using percent compositions we will also determine the empirical and molecular formula.
|
Experimentally determine the molecular formula of magnesium oxide.
Molecular Formula of Magnesium Oxide |
Day 6.9 3/22 |
Day 6.10 3/27 |
Day 6.11 3/29 |
|
Review: Today we will be reviewing for the upcoming exam. You will be required to complete 30 points worth of review activities.
Exam 6 and SLO Review Solutions to Exam 6 and SLO |
Exam 6
|
SLO
|