Enduring Understandings:
- The components that make up the atom determine the characteristics of that atom.
- The physical and chemical properties of matter give us clues about how its atoms or molecules are arranged and how they interact.
- Studying the changes in matter can help us understand what is happening on an atomic or molecular level.
- The physical and chemical properties of matter give us clues about how its atoms or molecules are arranged and how they interact.
- Studying the changes in matter can help us understand what is happening on an atomic or molecular level.
- Scientists express electron configuration differently for different purposes.
- The components that make up the atom determine the characteristics of that atom.
- The wavelength/frequency of light released/absorbed by an atom correlates to its atomic structure.
- Patterns of properties emerge from the arrangement of elements on the Periodic Table and give it predictive power.
Objectives
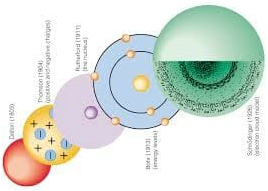
Atomic Theory: Know and be able to describe the contributions the following scientists have made to the atom and atomic theory.
ionic radius
electronegativity
ionization energy
electron affinity
- Democritus
- John Dalton: identify Dalton’s postulates and evaluate their accuracy in light of current atomic theory.
- JJ Thomson: explain how Thomson’s experiments led to his discovery of electron properties.
- Ernest Rutherford: explain how Rutherford’s experiments led to his conclusions about the nuclear atom.
- Niels Bohr: explain Bohr’s addition of electron energy levels to Rutherford’s nuclear atom.
- Erwin Schrodinger
- James Chadwick
- express the arrangement of all electrons in an atom using electron configuration
- Using the idea of quantum mechanics describe the areas of probability where an electron can be located
- From the electron configuration identify which electrons are valence electrons and how they are used in bonding
- describe the electromagnetic spectrum
- describe the mathematical relationships between energy, frequency, and wavelength of light
- calculate wavelength and frequency using the speed of light
- calculate energy and frequency using Planck’s constant
- explain how the contributions of scientists such as Mendeleev led to the development of the Periodic Table as a predictive tool
- explain how chemical and physical properties were historically used to develop the Periodic Table
- identify and explain properties of chemical families, including alkali metals, alkaline earth metals, halogens, noble gases, and transition metals
- use the Periodic Table to identify the chemical family of an element and explain its properties
- identify and explain periodic trends, including:
ionic radius
electronegativity
ionization energy
electron affinity
- Distinguish and identify between physical and chemical properties and changes
- Properties and Structure of Matter: be able to distinguish/identify:
- the difference between a pure substance and a mixture
- the difference between a homogeneous and heterogeneous mixture
- the phase changes and what is occurring at the molecular level to the atoms/compounds
- From a phase diagram be able to identify if a change in phase is endothermic or exothermic
- From a triple point curve be able to identify:
- where the phase changes occur and what phase a substance is at a specific temperature and pressure
- the triple point, boiling point and freezing point on the diagram
Atomic Theory Packet
Day 0: Winter Break |
Day 1: 1/4 |
Day 2: 1/9 |
|
The Story of Atom: Watch the following video about atomic theory and take notes. These notes may be used on the daily quiz given 1/4.
Dead Dudes Note Organizer You might also like to check the following below: The Story of Atom Crash Course: History of the Atom Just How Small is an Atom |
Properties of the Periodic Table: Today we will begin to look deeper into the element and discuss ions, isotopes, and determine how the mass on the table is calculated. We will also complete Organizing the elements.
Organizing the Elements Solutions to Lesson 4.1 |
The Movement of an Electron and It's Energy:
Electromagnetic Radiation and Energy PPT Solutions to Lesson 4.2 |
Day 3: 1/11 |
Day 4: 1/13 |
Day 5: 1/18 |
|
The Energy of Light: Today you will be conducting a flame test lab to help identify an unknown metal based on the color of flame it produces when burned. We will begin Quantum numbers and electron configuration.
Watch the following below to get ahead of the game |
How does the Arrangment of the Electrons determine Properties: In class continue discussing electron configuration, orbital notation. How does the specific arrangement determine physical and chemical properties. Determine valance electrons and write quantum numbers from orbital notation and electron configuration.
Solutions to Lesson 4.3 & 4.4 Quiz Review Solutions to Quiz Review Kahoots if interested https://play.kahoot.it/#/k/0545e417-c4eb-452d-9204-75004554c8a0 https://play.kahoot.it/#/k/e5de7f90-8b64-416b-89c5-601fa97520a8 https://play.kahoot.it/#/k/f08d69bc-043b-4b54-8e62-d7e4b6d97ba5 https://play.kahoot.it/#/k/311b0c03-3275-41e0-a1de-6242aa97d635 |
Day 6: 1/20 |
Day 7: 1/23 |
Day 8: 1/25 |
|
Quiz over days 1-4
Can you Determine the Density of Germanium and Flerovium: Using the properties of other elements you will determine the density of Germanium and Flerovium. Can you Determine the Density of Germanium and Flerovium Day 1:
|
Continue Can you Determine the Density of Germanium and Flerovium: Using the properties of other elements you will determine the density of Germanium and Flerovium.
Day 2:
|
Finish Can you Determine the Density of Germanium and Flerovium: Using the properties of other elements you will determine the density of Germanium and Flerovium. Use the template shared with you in google drive.
Day 3:
Please watch for next class. Periodic Trends Lecture Video |
Day 9: 1/30 |
Day 10: 2/1 |
|
Can You Determine a Property of an Element Based on its Position? Today we will look at how the properities of elements repeat themselves in perdictable manners. Specifically atomic radius, ionic radius, ionization energy, and electronegativity.
Properties of Periodic Table and Periodic Trends PPT Unit 4 Concept Sheet Unit 4 Review Unit 4 Review Answers |
Exam 4
|
Class Handouts |
Useful Links |